
1444 Rainville Road, Tarpon Springs, FL 34689, U.S.A.
Tel. +1-727-940 3944; Fax +1-727-934 9275; Email:
basicresearch(at)i-b-r(dot)org
Main Page | | IBR Grants | IBR Conferences |
Latest Discoveries
About Us | CV of IBR Members |
OPEN ETHICAL COMPLAINT AGAIST MR. JOSEPH M. CALO
OF BROWN UNIVERSITY WITH REQUEST OF
TERMINATION OF EMPLOYMENT
filed by
Dr. Ruggero Maria Santilli
Curriculum
35246 US 19 No. # 215, Palm Harbor, FL 34684
Tel. 727-688 3992, Fax 727-934 9275, Email basicresearch@i-b-r.org
to
Dr. Christina Paxson
Office of the President
Box 1860, 1 Prospect Street, Providence, RI 02912
Phone 401-863-2234, Fax 401-863-7737, Email: president@brown.edu
Dr. Lawrence E. Larson
Dean of Engineering
School of Engineering Brown University, Box D 184 Hope Street, Providence, RI 02912
Phone 401-863-2677, Fax 401-863-1238, Lawrence_Larson@brown.edu
with copy to
Mr. J. M. Calo
Division of Engineering, Box D, Brown University, Providence, RI 02912, USA
Tel.: +1 401 863 1421; fax: +1 401 863 9120. E-mail address: JMCalo@brown.edu
The Open Ethical Ccomplaint is availabnle in the website
http://www.i-b-r.org/J-M-Calo-Denunc.htm
SUMMARY
by Luca Petronio
Editor according to Wikipedia rules of the
Contributions to this blog should be sent to the email luca@scientificethics.org
The Italian-American scientist R. M. Santilli (Curriculum) has recently filed an Ethical Complaint against J. M. Calo in his capacity as engineer of Brown University, Providence, R.I. Santilli has filed the Ethical Complaint in his capacity as pro se stockholder of the U. S. public company Magnegas Corporation (whose stock is traded at NASDAQ under the symbol MNGA) on grounds of emotional distress and sizable financial losses caused by Calo in various dishonest claim on the technology underlying said company, that of Santilli new chemical species of magnecules and its various industrial applications, all misconducts having been perpetrated under Calo's formal affiliation to Brown University. Santilli request the termination of employment of J. M. Calo by Brown University and other reparatory measures. Some of the main aspects of the highly detailed Complaint requiring post Ph. D. knowledge in mathematics, physics and chemistry, can be summarized as follows.
In the experimental paper entitled "A new gaseous and combustible form of water," published in the Intern. J. Hydrogen Energy 31, 1113 (2006),
Ref. [6] of the Complaint , R. M. Santilli presented a number of measurements following signed reports by qualified U. S. analytic laboratories showing the presence of chemical anomalies in a gas called by Santilli HHO, but sold by Hydrogen technology Applications, Inc., under the commercial name of Aquygen and produced via a patented electrolyzer of distilled water .
Among various remarkable anomalies, Santilli presented in Ref. [6] analytic documentation that:
1) The HHO gas instantly melts bricks and tungsten at contact without any need of atmospheric oxygen, a thermal energy transfer which is known to be
impossible for a stochiometric mixture of 2/3 hydrogen and 1/3 oxygen;
2) The existence of anomalous chemical species in the HHOO gas in the range from 3 amu to hundreds of amu established by numerous chromatographic analyses, which anomalous species cannot be identified among known molecules and do not have the IR signature "at their amu value " (rather than those of the cluster constituents), necessary for a credible interpretation the anomalous species as conventional molecules;
3) An anomalous adhesion of the HHO gas to various substances, including anomalous macroscopic peaks in the MS blacks following scans and flushing under the verification of no anomalies of the blanks prior to the scans, which feature has an evident environmental relevance suggested for study; and other anomalies.
Soon after the appearance of Santilli's paper [6], the "engineer" Mr. Joseph M. Calo published a litany of "criticisms"
Ref. [33] of the complaint below
under his formally stated affiliation to Brown University of Providence, R.I., claiming that all the anomalies of the HHO gas documented in paper [6] do not exist. Calo then
a) presented a never ending show of old 20th century knowledge none of which had
any connection whatsoever with the treated topics in ref. [6] and, in fact, without even attempting the use of old knowledge for a quantitative interpretation of the anomalous features of the HHP gas apparently due to its known impossibility;
b) accuses serious U. S. laboratories of misconducts in the release of signed reports, under which claim the laboratories could lose their license with ensuing possible lawsuits by said laboratories against Brown University;
c) dubs as "pseudoscience" the magnegas technology at the core of operation of the U. S. public company Magnegas Corporation thus clearly damaging the value of its stock traded at NASDAQ under the symbol MNGA, with additional potential lawsuits by stockholders of Magnegas Corporation against Brown University;
d) discredits the industrial development, following the separate private investment of millions of dollars by the petroleum industry, of environmentally clean fuels baed on Santilli's magnecular bond between hydrogen and fossil fuels, said magnecular bond being the sole known for a stable bond of the gaseous hydrogen to liquid or solid petroleum fuels, said discredits
implying consequential potential attack against Brown University by environmental groups due to the transparent environmental, industrial and societal damages caused by Mr. Calo; and
e) ventures numerous additional simply irresponsible claims which can only be understood as originating from a fanatic fervor to oppose Santilli's discoveries without any provocation, advance notice or prior verifications.
Immediately following the appearance of "criticisms" [33] by the "engineer" J. M. Calo, the "Professor of Chemistry" M. O. Cloonan from Ireland released a detailed rebuttal,
Ref. [34] of the complaint below, and the "Professor ofChemistry" (the late) Jerdsay V. Kadeisvili from Georgia Russia, released his additional rebuttal
Ref. [35] of the complaint below, both rebuttals being published in the same journal of the original Santilli paper [6] and of Calo's "criticisms" [33].
Independent rebuttals [34,35] by the "Professors of Chemistry"" (not engineers) M. O. Cloonan and J. V. Kadeisvili constitute an integral part of Santilli's Ethical Complaint against J. M. Calo of Brown University in which Santilli additionally provides documentation that:
A) Calo's "criticisms" [33] against Santilli's paper ]6] are pompous, arrogant, and deprived of any scientific content whatsoever, thus being highly unethical and in gross violation of the Ethical Rules of Brown University (sections 1 and 2 of the Ethical Complaint);
B) Calo's "criticisms" [33] are fully in line with the opposition Santilli suffered in the early 1980s when he was at the Department of Mathematics of Harvard University under formal DOE contracts. Said opposition was perpetrated by Harvard's employees of the time Steven Weinberg, Sheldon Glashow and Sidney Coleman and was intended to prevent, disqualify and disrupt DOE plans on the development of a time-irreversible Lie-admissible covering of 20th century mathematical, physical and chemical theories for consistent treatment of energy releasing processed (that are notoriously irreversible over time). these studies implied
a necessary covering of Einstein's theories that was fanatically opposed by said Harvard employees without any sense of dignity or shame (see Section 3 of the Ethical Complaint and
the 1984 Ethical Probe of Einstein Followers in the USA: An Insider's view, that includes three volumes of documentation and the supporting editorial of The Harvard Crimson dubbing these unethical conducts as The Politics of Science, 2007 denunciation Update and the
Denunciation of the Gangsterism by the physicist Steven Weinberg);
C) Unethical "criticisms [33] by the "engineer" J. M. Calo were immediately reproduced by Arthur L. Rubin of Wikipedia in Santilli's article
https://en.wikipedia.org/wiki/Ruggero_Santilli,
while Rubin systematically prohibited the quotation in Santilli's article of the rebuttals by "Professor of Chemistry" M. O Cloonan form Ireland [34] and "Professor of Chemistry" Jerdsay V. Kadeisvili from Georgia Russia [35].
Additionally, Rubin prohibited the quotation in Santilli's article of papers [17,18] of the Ethical Complaint below co-authored by the "Professor of Chemistry" at Virginia Commonwealth University Don Shillady; dubbed for over a decade as "fringe the technology of the U. S. public company Magnegas Corporation" and perpetrated systematic abuses of his authority as editor of Santilli's page in blatant violation of Wikipedia rules.
Santilli's Ethical Complaint provides evidence of an organized anti-American conspiracy by a hidden sinister cartel in the academic-Wikipedia complex which has been conceived, organized and maintained under the abuse of billions of dollars of taxpayers money to oppose, disrupt and disqualify undesired basic advances in chemistry beyond the old. 20th century, quite limited capability of quantum chemistry, the misconducts of the cartel in other scientific fields being denounced elsewhere, resulting in the suppression of even the shadow of scientific democracy for qualified inquiries in the United States of America (as one sample of misconducts among too many for confront, see the Denunciation of organized Scientific Crime in the U.S chemistry.
Santilli's Ethical Complaint requests to Brown University administrators of terminating the employment as well as any direct or indirect affiliation of J. M. Calo to Brown University as a necessary condition for Brown University to maintain its otherwise excellent reputation, and concludes with an appeal to serious scientists as well as to American people to stop the current complicity via silence in the ongoing organized scientific crime in the U.S.A. and organize a systematic disqualification of the ongoing scientific conspiracy in which absence Santilli feels obliged to recall that "As set in history, people have the destiny they they want or deserve".
Luca Petronio
Editor according to Wikipedia rules
Contributions to this blog should be sent to the email luca@scientificethics.org
========================================
OPEN ETHICAL COMPLAINT AGAIST MR. JOSEPH M. CALO
OF BROWN UNIVERSITY WITH REQUEST OF
TERMINATION OF EMPLOYMENT
filed by
Dr. Ruggero Maria Santilli
Curriculum
35246 US 19 No. # 215, Palm Harbor, FL 34684
Tel. 727-688 3992, Fax 727-934 9275, Email basicresearch@i-b-r.org
to
Dr. Christina Paxson
Office of the President
Box 1860, 1 Prospect Street, Providence, RI 02912
Phone 401-863-2234, Fax 401-863-7737, Email: president@brown.edu
Dr. Lawrence E. Larson
Dean of Engineering
School of Engineering Brown University, Box D 184 Hope Street, Providence, RI 02912
Phone 401-863-2677, Fax 401-863-1238, Lawrence_Larson@brown.edu
with copy to
Mr. J. M. Calo
Division of Engineering, Box D, Brown University, Providence, RI 02912, USA
Tel.: +1 401 863 1421; fax: +1 401 863 9120. E-mail address: JMCalo@brown.edu
Dear Drs. Paxson and Larson
The underwriter Ruggero Maria Santilli, acting as a pro se stockholder of the American public company Magnegas Corporation (whose stock is traded at NASQAD under the symbol MNGA, www.magnegas.com), hereby requests that Brown University examines this Ethical Complaint against your employee Mr. J. M. Calo and terminates his employment on grounds identified and documented below of excessive violations of scientific ethics and accountability, excessive scientific corruption on the technology of a publicly traded company, excessive obstructions against environmental fuels under development by the petroleum industry, excessive lies against highly qualified and reputable analytic laboratories in the U.S.A., excessive damage to the image of the American science in the world as well as in history, all misconducts perpetrated via the abuse of the credibility of Brown University whose image will be permanently tarnished in the event of any continuation of any affiliation of Mr. J. M. Calo to your otherwise beautiful and renowned college.
This complaint is filed on grounds of the Ethical Rules of Brown University as available in the website
https://secure.ethicspoint.com/domain/en/report_company.asp?
clientid=25461&override=yes&agreement=no&violationcategoryid=3173
with particular reference to:
A. Fraud - Research
The intentional misrepresentation or concealment of information in order to deceive, mislead, or acquire something of value. A fraud is an intentional deception perpetrated to secure an unfair gain. (Examples include: Violation of Policy, Sabotage).
B. Intellectual Property Infringement, Misappropriation or Disclosure
Any unauthorized or inappropriate use, misappropriation or disclosure of confidential information (in any form) or intellectual property belonging to the institution or any institution's customer, supplier or business partner, including, without limitation, any intellectual property protected under any U.S. or other laws relating to copyrights, patents or trade secrets. Also includes any unauthorized or inappropriate use of any institution computer system.
C. Research Misconduct
Fabrication, falsification, plagiarism, or other practices that seriously deviate from those that are commonly accepted within the scientific community for proposing, conducting, or reporting research. Fabrication is making up data or results and recording or reprinting them. Falsification is manipulating research materials, equipment or processes, or changing or omitting data or results, such that the research is not accurately reported in the research record. Plagiarism is the appropriation of another persons ideas, processes, results, or words without giving appropriate credit.
The above rules are hereinafter referred to as "Ethical Rules A, B. C of Brown University."
1. SCIENTIFIC BACKGROUNDS
I here reproduce verbatim Section 1 of the experimental paper [25] published in a refereed journal by independent scientists:
In memoir [1] of 1998, the Italian-American physicist R. M. Santilli (curriculum) presented for the first time mathematical, theoretical and experimental evidence on the existence of a new chemical species today known as Santilli magnecules which is composed of individual atoms (H, O, C, etc.), dimers (HO, CH, etc.) and ordinary molecules (H_2, CO, H_2O, etc.) bonded together by attractive forces between opposing magnetic polarities of toroidal polarizations of atomic orbitals, as well as the polarization of the magnetic moments of nuclei and electrons (see a conceptual rendering in Fig. 1).
Santilli suggested for the new species the name "magnecules" in order to distinguish the new species from conventional "molecules" (namely, clusters of atoms under the conventional valence bond), as well as to indicate the primary magnetic origin of the new bond. The symbol "-" is widely used to denote a valence bond (such as H-H) while the symbol "×" is used to denote a magnecular bond (such as H×H).
The main theoretical argument of Ref. [1] is that the toroidal polarization of the electron orbitals creates a magnetic field (due to the rotation of the electrons within said toroid) which does not exist for the same atoms when the electron orbitals have the conventional spherical distribution. When two so polarized atoms are at a sufficiently close distance, the resulting total force between the two atoms is attractive because all acting forces are attractive except for the repulsive forces due to nuclear and electron charges. However, the latter forces can be averaged to zero in first approximation since the individual atoms have a null total charge. Alternatively, individual atoms can be assumed in first approximation to have a null total charge distribution, resulting in the evident dominance of the attractive magnetic forces between two atoms with toroidal polarization of their orbitals.
Memoir [1] then provides means for the actual creation of a gas with the new magnecular chemical structure. In essence, Santilli recalled the well known property that the polarization of the electron orbitals from their natural spherical distribution to the needed toroidal form requires extremely high magnetic fields (expected to be of the order of 10^{10} Gauss or more) that, as such, are not available in our macroscopic environment.
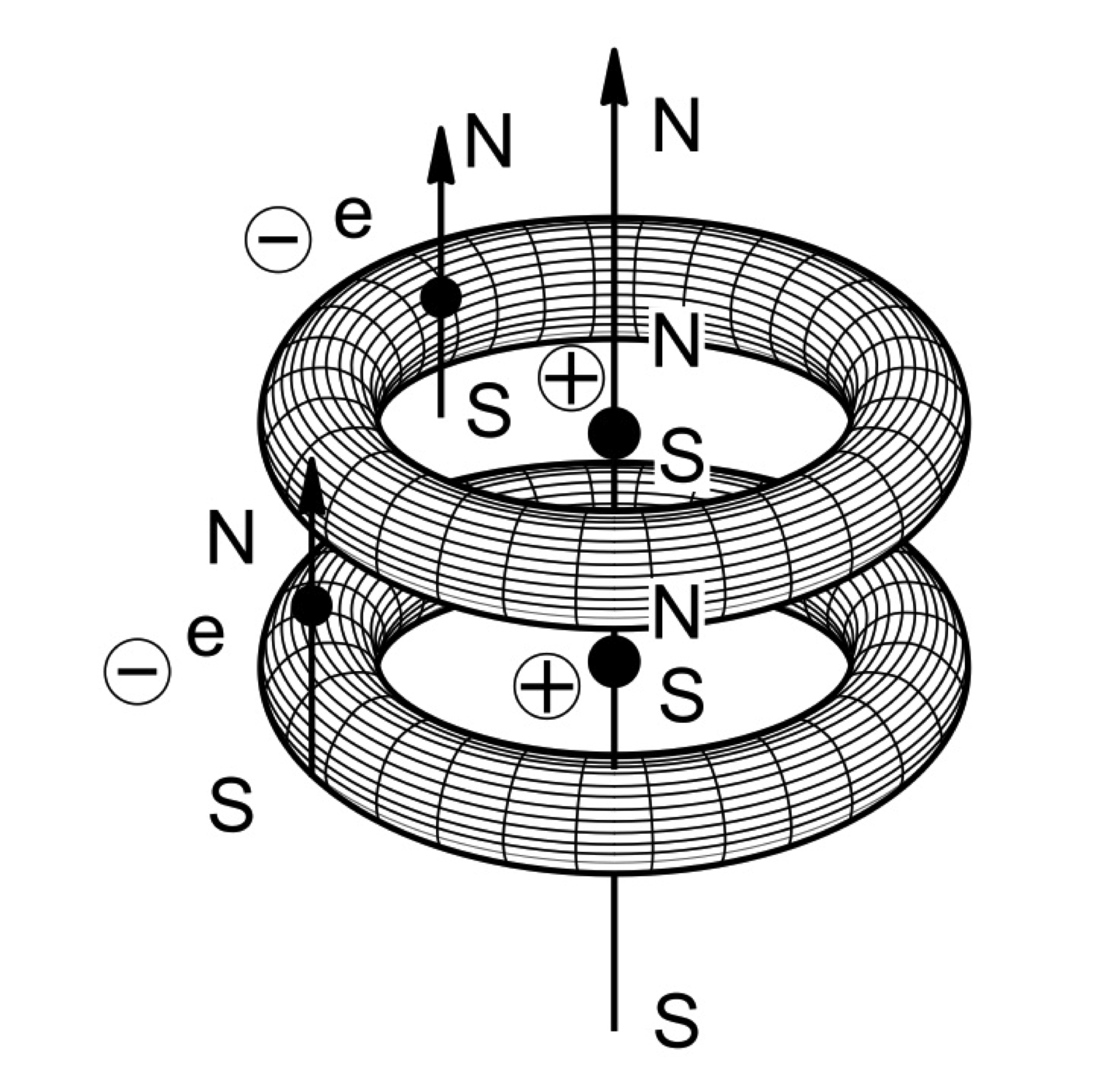
Fig. 1. Conceptual rendering of Santilli magnecules, here referred to the species H×H assumed at absolute zero degree temperature. Note that: the toroidal polarization of the electron orbitals creates a new magnetic field not existing for spherical distributions; all magnetic forces between said toroids as well as between the magnetic polarities of nuclei and electrons are attractive; and the repulsive forces between nuclear and electron charges can be averaged to zero in first approximation since the atoms are individually neutral, thus resulting in the dominance of a new non-valence bond Santilli calls ``magnecular." Note that the new bond can be also called ``axial" (or ``perpendicular") in the sense that the magnetic force exists along the symmetry axis of the toroid (or the magnetic force is perpendicular to the symmetry plane of the toroid).
Therefore, for the creation of the new magnecular species, Santilli suggested in memoir [1] the use of a DC electric arc between graphite electrodes submerged within a liquid (such as distilled water). As it is well known, the arc decomposes the liquid molecules into mostly ionized atoms by creating between the tip of the electrodes a plasma composed of H, C, and O individual atoms, CH and OH dimers, and ordinary molecules such as H_2, CO H2O and others. Santilli then noted that at atomic distance from said electric arc, the magnetic field has indeed the desired strength since said magnetic field is inversely proportional to the distance (of the order of 10^{-8}~cm) and directly proportional to the electric current (of the order of 10^3 A or more), thus having a strength of the order of 10^{11} Gauss which is sufficient to achieve the desired toroidal polarizations of the electron orbitals (see the conceptual rendering of Fig 2).
Additionally, the strong magnetic field surrounding a DC arc naturally aligns polarized atoms in the needed sequence of magnetic polarities South-North-South-North, etc. resulting in the configuration of Fig. 1. It is evident that, as soon as the arc is disconnected, atoms return to their natural spherical distribution due to collisions and other reasons. Santilli's main chemical argument is that the spherical distribution is indeed recovered but for the bonded pairs of polarized atoms as in Fig. 1, since said spherical distribution cannot be achieved for each individual atoms of the bonded pair due to insufficient energies to break said bond.

Fig.2. Conceptual rendering of Santilli's production of a gas with magnecular structure via the use of a submerged electric arc that, at atomic distance, is sufficiently powerful to polarize electron orbitals into toroids and then align polarized atoms essentially along the configuration of Fig 1.
Memoir [1] then presented a considerable experimental evidence on the existence of the new species of magnecules for a gas created via the above reviewed method. The verification (reviewed in more details in Section 2) was achieved via the use of a GC-MS/IRD, namely, a Gas Chromatographer Mass Spectrometer equipped with an InfraRed Detector. Santilli's main experimental argument is that all gas chromatographic equipment available in the late 1990s had been conceived and established for the detection of conventional molecular species.
The only possibility available at the time of memoir [1] to ascertain the existence of the new species of magnecules was that of subjecting the same injection, first for detection via the GC-MS and then detection via the IRD. The identification of clear clusters in the GC-MS that have no IR signature establishes the existence of magnecules since their bond is stable at ambient temperature, but weaker than the valence bond, thus having no IR signature. The magnecular nature of the bond was then confirmed by the arc method for its creation. Santilli insisted that it was impossible to achieve the same results in a resolutory way via two separate instruments due to the impossibility of matching scans in the GC-MS with scans in a separate IRD without any ambiguities.
Gas chromatographic analyses reported in memoir [1] were conducted via a GC-MS/IRD consisting of a HP GC model 5890, a HP MS model 5972, and a HP IRD model 5965 operated in rather unusual conditions described in details in ref. [1], such as: largest available feeding line of at least 0.3 mm ID; cryogenic cooling of the feeding line; lowest available column temperature of 10^0 C; longest available elusion time of about 25 minutes; and other conditions. Representative chromatographs out of a considerable number of scans of memoir [1] are reported in Figs. 3-5. Memoir [1] also reported the confirmation of the results (here not reproduced for brevity) obtained via an identical GC-MS/IRD located at a different laboratory.
Memoir [1] also provided considerable experimental evidence for the existence of magnecules in liquids, and comments on the expected existence of magnecular bonds in solids.
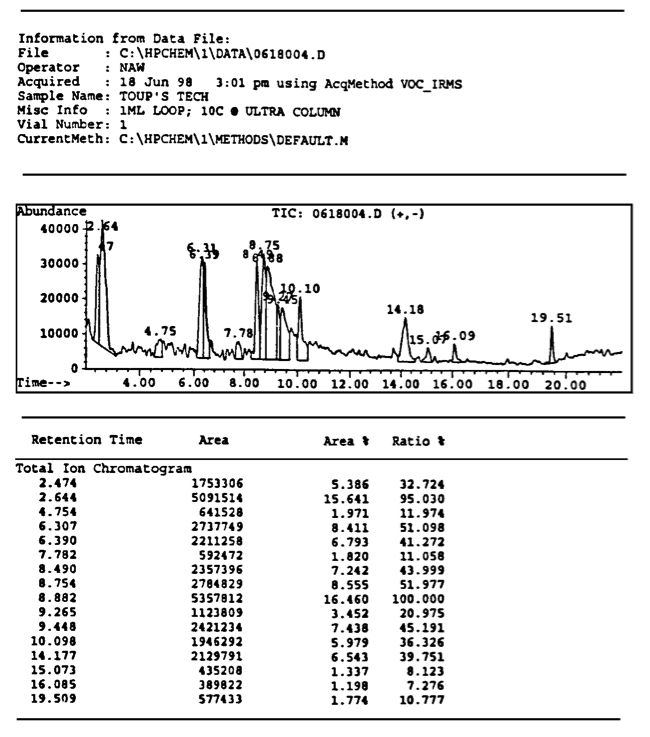
Fig.3. A reproduction from memoir [1] of the GC-MS peaks between 40 and 400 a.m.u. on a gas with a magnecular structure obtained on June 19, 1998, by analysts Louis A. Dee and Norman Wade of the branch of National Technical Systems (NTS) located at the McClellan Air Force Base in North Highland, near Sacramento, California. The test was done on a type of magnegas produced via an electric arc between graphite electrodes submerged in tap water. Therefore, only the CO_2 peak was expected to appear in the scan in a macroscopic percentage, while no dominant CO_2 peak is visible in the scan.
Following the appearance of memoir [1], M. G. Kucherenko and A. K. Aringazin [2] confirmed in Ref. [2] of 1998 the numerical value computed by Santilli for the magnetic field along the axial symmetry of a toroidal polarization of the electron orbital of the Hydrogen atom as being about 1,347 times stronger than the nuclear magnetic field, thus confirming the presence of a magnetic field sufficiently strong to create a bond of the type of Fig. 1.
A. K. Aringazin conducted in Ref. [3] of 2001 an in depth theoretical analysis of the strong magnetic fields needed to achieve the toroidal polarization of electron orbitals, thus confirming Santilli's use of a submerged DC electric arc in order to achieve a magnetic field of the needed strength.
In 2001, Santilli released monograph [4] on a systematic presentation of the status of the knowledge in the new field in early 2000. In particular, magnecules were presented in their proper technical environment, that of a non-unitary covering of quantum chemistry nowadays known under the name of isotopic branch of hadronic chemistry orisochemistryf for short. Monograph [4] also detailed the first industrial realization of the the new species of magnecules consisting of the gaseous fuel produced and sold world wide under the trade name of MagneGas^{TM} (MG) by the U. S. company Magnegas Corporation which is publicly traded at NASDAQ under the symbol MNGA (see www.magnegas.com for details). This application essentially consists of the gasification of liquids via a submerged electric arc into the gaseous fuel MagneGas that possesses anomalous features, such as a combustion flame temperate double that of natural gas, metal cutting faster than acetylene, combustion exhaust without hydrocarbons thanks to a full combustion (because the magnecular bond is weaker than the valence bond), and other features.
Santilli presented in Ref. [5] of 2003 theoretical and experimental evidence for the second industrial application of the new species of magnecules, consisting of the synthesis of ``heavy Hydrogen and Oxygen," nowadays denoted with the chemical symbols MH and MO, respectively, and consisting of magnecular clusters of Hydrogen or Oxygen atoms, respectively. When detected via GC-TCD equipment operated at high temperature (so as to destroy the magnecular bond), MH and MO can be constituted by conventional Hydrogen or Oxygen up to 99% pure, yet their specific weight is a multiple that of conventional molecular Hydrogen and Oxygen.
The new species MH and MO were presented in Ref. [5] as evidence on the very existence of magnecules since the Hydrogen has only one valence electron to share and, as such, cannot possibly achieve valence bonds for more than two Hydrogen atoms (see the forthcoming paper [16] for details).
Santilli then provided in Ref. [6] of 2006 theoretical experimental evidence on a third industrial application of the new species of magnecules, here referred to the gas commercially produced via certain electrolyzers and essentially consisting of 2/3 Hydrogen and 1/3 Oxygen, which contains a small percentage of H and O magnecular clusters. Under these conditions, Santilli suggested in Ref. [5] the name of HHO (although a similar gas produced via a different electrolyzer is known as Brown's gas).
M. O. Cloonan presented in Refs. [7-9] of 2006 to 2009 applications of Santilli magnecules and its underlying hadronic chemistry to particular forms of pericyclic reactions and related new structures, thus illustrating the expected capability of the new species of Santilli magnecules to produce new chemical substances (i.e., chemical substances not entirely based on valence bonds).
In monograph [10] of 2008, R. M. Santilli presented an update on the first industrial application of the new species of magnecules via the combustible gas MagneGas indicated earlier, as well as an update of the additional industrial applications for MH, MO, and HHO.
In Ref. [11] of 2011, R. M. Santilli presented the industrial application for which he developed the new species of magnecules, namely, their use as a necessary premise for the achievement of nuclear fusions without harmful radiation. Santilli's main argument is that preceding attempts at achieving systematic nuclear fusions were flawed by an inability to systematically control the exposure of nuclei out of their electron clouds, the systematic and controlled proper alignment of nuclear spins and other insufficiencies. Inspection of the very conception of a magnecules as in Fig 1 illustrate the resolution of these problematic aspects in a systematic, repeatable and controlled way.
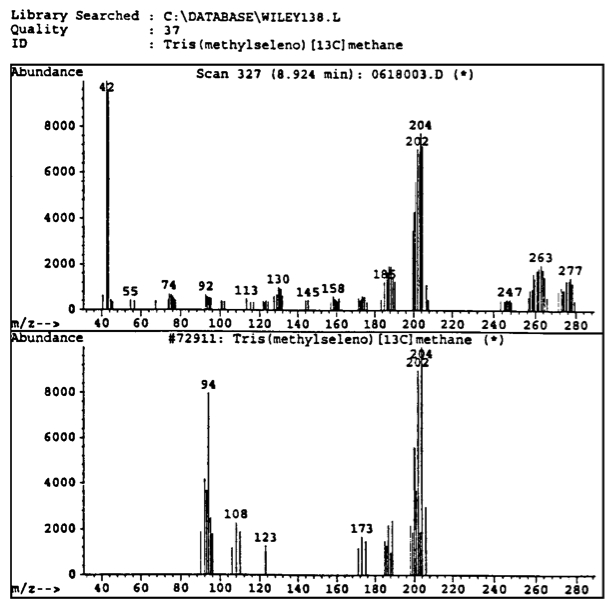
Fig. 4. A representation (top view) from memoir [1] of the first experimental evidence at NTS laboratories that the peaks of Fig. 3 are ``unknown" because of a lack of identification by the computer among 500,000 known molecules. The same situation occurred for all remaining peaks of Fig. 3. A reproduction (bottom view) from memoir [1] on the scan of Fig. 3 obtained via the IRD component of the GC-MS/IRD providing the first experimental evidence on the lack of IR signature of the GC-MS peaks of Fig. 3. In fact, the above IRD scan establishes the existence of large clusters that have no IR signature at all, thus dismissing valence bonds since they would require perfectly spherical clusters that are essentially impossible at large a.m.u. The only identified IR signature is that for the conventional molecule CO_2, but the latter refers to the constituents of the peaks of Fig. 3, because it is not identified as an isolated detectable species.
The nuclear physicists Robert Brenna, Theodore Kuliczkowski, and Leong Ying presented in Ref. [12] of 2011 an independent verification on the existence of Santilli's new fusions without harmful radiation, and confirmed in particular via various chemical analyses the necessary use of magnecular structures for their achievement.
In Ref. [13] also of 2011 Santilli presented additional cases of nuclear fusion, this time for Silica, based on the use of a magnecular bond of the type of Fig. 1 between Carbon and Oxygen, plus a "trigger," namely, a mechanism pushing nuclei at 1 fm mutual distance at which the strongly attractive nuclear forces are activated and fusion becomes inevitable.
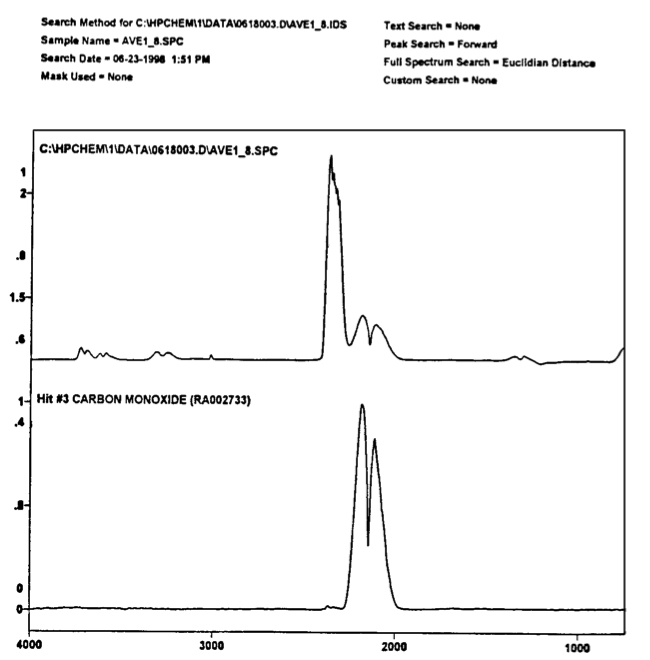
Fig. 5. Reproduction from memoir [1] of the first experimental evidence on the mutation of the IR signature of molecules under a strong magnetic field, here referring to the CO_2 (top) compared to the result of the computer search (bottom). Note that there is the appearance of two anomalous IR signature suggesting new internal bonds, and that the computer erroneously interpreted the molecule as being CO rather than CO_2.
To complete the references on Santilli magnecules, we should additionally quote the second edition of monograph [14] where Santilli presents the application to biology of the new mathematics of increasing complexities known under the names of Santilli iso-, geno-, and hyper-mathematics. In this monograph, Santilli also shows that most biological structures, particularly complex structures such as the DNA, cannot have a molecular structure due to an insufficient number of valence electrons to bond together an extremely large number of atoms. Santilli has introduced the notions of Santilli IsoMagneCules, GenoMagneCules, and HyperMagneCules (also presented in Ref. [4]) to initiate deeper studies on the extreme complexity of biological systems.
In the recent signed report [15]., the Eprida Laboratory of Atlanta, Georgia, achieves an experimental confirmation of Santilli MagneHydrogen (MH) via measurements showing 98% H-purity while having 3.4 times the specific weight of conventional H2.
In the recent recently published paper [16], the authors present the second independent experimental confirmations of Santilli's new chemical species of MagneHydrogen and MagneOxygen (MO) originally presented in Ref. [5], which constitute some of the best evidence on the existence of magnecules, and confirm the anomalous values of their specific weight, energy content and other features.
In Ref. [17], Santilli and Don Shillady (Professor of Chemistry at Virginia Commonwealth University) provides the first known numerically exact representation of the binding energy and other features of the conventional H_2 molecule via unadulterated first principle of the isotopic branch of hadronic chemistry [4]. This paper also provides the first known quantitative formulation of the conventional valence bond represented by an equation characterizing an "attractive" force between valence electrons in single coupling compatible with experimental data. To understand this result, one should keep in mind that the formulation of the valence bond via quantum chemistry cannot achieve an explicit expression of an attractive valence force since identical electrons are predicted to repel (and certainly not attract) each other according to quantum mechanics.
In Ref. [18] Santilli and Shillady achieve the first known quantitative, exact representation of the binding energy and other features of the water molecule via unadulterated first principle of the isotopic branch of hadronic chemistry, Refs. [19,20] are some of independent works on the Santilli-Shillady structure model of the H_2 and water molecules (see also monograph [4] for a review).
The work herein presented has been reviewed in Refs. [22-23], while a general review of the mathematical, physical, and chemical aspects underlying the new species of magnecules has been provided by I. Gandzha and J. V. Kadeisvili in monograph [24]. This concludes the identification of all published references on Santilli magnecules to to the authors best knowledge at the time of writing this paper (June 2013).
This paper is specifically devoted to the presentation of additional experimental confirmations of the new species of Santilli magnecules as presented in the original memoir [1] of 1998. In the hope of stimulating a collegial participation in this new field of chemistry, the authors will make available to qualified chemists at no cost samples of gases with magnecular structure under the availability of gas chromatographic equipment suitable for the needed measurements.
In Sect. 2 of Ref. [25], following the presentation of vast experimental evidence I cannot possibly reproduce here, Drs. . Yang, J. V. Kadeisvili, and S. Marton state:
Following initial doubts due to their novelties under consideration, our analyses confirm all of Santilli's claims presented in memoir [1] for the case of gases with magnecular structure without exceptions known to the authors. Out of a large variety of tests with different magnecular gases, different equipment and different procedures, our main results are presented in the representative scans of in Figs. 6 to 19, and can be summarized as follows:
1. Characterization of magnecules by weakly bonded individual atoms, dimers, and conventional molecules;
2. Stability of magnecules at ambient temperature;
3. Progressive reduction of magnecules with the increase of the temperature;
4. Termination of magnecules at a suitable Curie temperature;
5. Detection of magnecular clusters under a suitably selected and operated GC-MS;
6. Transparency of magnecules to infrared detectors for the a.m.u. of the clusters (and not at a smaller a.m.u. characterizing constituents);
7. Dependence of detected magnecules from the elusion time;
8. Dependence of magnecular species from filtration and compression;
9. Anomalous adhesion of magnecular gases to disparate materials;
10. Anomalous mutation of magnecular clusters in time and under different detection procedures or equipment;
11. Anomalous accretion of magnecular clusters by individual atoms; and other features.
Among over thirty international conferences organized in the field in the past three decades, I quote the 2013 international Conference on Hadronic Chemistry in India [26]; my keynote talk at that conference [27]; and the forthcoming conferences on the novel Santilli isomathematics underlying the new chemical species of magnecules [29,30].
The mathematical, theoretical, experimental and industrial validity on Santilli magnecules has been documented by hundred of certifications, independent statements and news releases by the U. S. public company Magnegas Corporation as available in the website of the company
http://ir.stockpr.com/magnegas/company-news. Additional news releases have been provided in Refs. [31-32].
2. DOCUMENTATION OF ETHICAL MISCONDUCT BY MR. J. M. CALO
AT BROWN UNIVERSITY
Soon after the publication of my paper [6] on "A new gaseous and combustible form of water," published in the Intern. J. Hydrogen Energy 31, 1113 (2006), without any provocation, advance notice or contact with me, Mr. Joseph M. Calo ("Calo" hereinafter) published "criticisms" [33] of paper [5] under his formal affiliation at the Division of Engineering, of Brown University, Box D, Brown University, Providence, RI 02912, USA.
In view of the complete lack of provocation, advance consultation or prior contact by Mr. Calo with me prior to his public "criticisms," it has been a question of principle for me to behave toward Mr. Calo in exactly the same way as he did with me, by therefore releasing these open ethical complaint against him without any advance consultation or contact with him.
Immediately following the appearance of "criticisms" [33], Martin O. Cloonan, Professor of Chemistry (rather than "Engineering") at the Chemistry, School of Science, Galway-Mayo Institute of Technology, Dublin Road, Galway, Ireland, published his rebuttal [34] in the same journal of Mr. Calo's criticisms [33].
The late Jerdsay V. Kadeisvili from Georgia, Russia, Honorary Professor of Chemistry at the Institute for Basic Research in Florida, published his additional rebuttal [35] also in the same journal of Mr. Calo's "criticisms" [33].
Profs. Cloonan and Kadeisvili rebuttals [34,35] essentially show that Calo's "criticisms" are unethical because:
1) The HHO gas was and continues to be industrially produced and sold in the U.S.A. by Hydrogen Technology applications Inc. (http://www.hytechapps.com/) under the commercial name of Aquygen. Similar gases are produced and sold by other companies under the name of Brown Gas. Therefore, any serious scientist can obtain the gas (generally available free of charge for tests) and verify the anomalous measurements reported in paper [6] following signed reports by highly qualified laboratories prior to the venturing of unethical judgments, or dismiss the measurements presented in paper [6] with counter-measurements.
2) Any serious scientist could have verified in person that the HHO gas melts brick and tungsten instantly at contact without any need for atmospheric oxygen, and verify in person that such an instantaneous melting of bricks and tungsten is impossible for a stoichiometric mixture of H2 and O2, thus establishing the chemical anomalies of the HHO gas beyond any possible or otherwise credible doubt and, at the same time, establishing Calo's ethical misconduct.
3) Any serious scientist could have personally verifies the numerous anomalous stable species in existing in the MS spectrum of the HHO gas and personally verify that such anomalous MS species admit no IR signatures as necessary to venture a molecular interpretation. Unfortunately for Mr. Calo, Brown University is a magnificent college with some of the most advances analytic equipment, including the latest GC, GC-MAS, IRD and other gas chromatographic detectors. Hence, it would have been quite easy for Mr. Calo to secure free samples of the HHO gas and re-run all MS and IR spectral analyses. Instead, Calo did not follow this standard scientific process in a premeditated way and proffered all sort of unethical statements intent to dismiss the claims of paper [6] via the abuse of the credibility of Brown University that, has carried the full responsibility of the occurrence.
Rebuttals [34,35] of Calo's misconduct [33] are an integral part of this Ethical Complaint and Request for Disciplinary Actions and they are not reproduced here for brevity. Instead, I provided below my analysis of the ethical misconduct by Mr. Calo via the quotation and
examination of each and every part of fraudulent claims [33] as follows:
The author of "A new gaseous and combustible form of
water," (Int. J. Hydrogen Energy 2006:31(09);1113�1128)
invites ... the "chemistry community for an independent
verification (of) . . . the HHO gas . . ." The following comments
are submitted in this spirit.
Mr. Calo's ethical misconduct starts in the above opening statement because I specifically requested independent technical appraisals and comments from the "chemical community" and not from a member of the "engineering community," due to the post Ph. D. character of the study in mathematics, physics and chemistry. In my fifty years of research and publications I have acquired the reputation of soliciting technical comments on any paper I have written prior to publication, and expressed my appreciation when my view was technically proved as being wrong. However, people would need medical examination in the event they dream that I can tolerate with grace irresponsible public attacks for clear political aims.
Anyone seriously attempting to establish the existence of any
new form of matter, such as "HHO gas" is claimed to be, especially
in a mixture of such common gases as hydrogen, oxygen,
and water vapor, would have to be extremely meticulous and
detailed in the appropriate selection and application of analytical
techniques, and in the interpretation of the resultant data,
in order to have any hope of convincing the scientific community.
However, as presented, the author demonstrates that he
does not have an elementary grasp of the analytical techniques
that were used, and/or the proper interpretation of the resultant
data.
The above statement by Mr. Calo is highly unethical because in paper [6] I present a number of completely standard measurements by quoting the used methods and identifying the laboratory by name and location. Expectedly being an academician, Mr. Calo knows that redundant information is rejected by serious editors. Above all, Mr. Calo's ethical misconduct becomes transparent from the fact that the above statement is studiously generic, namely, to act ethically, Mr. Calo should have identified at least "one" specific deficient measurement and identified at least "one" technical insufficiency. He did not, thus showing the premeditated intent to abuse the credibility of Brown University to oppose basic advances not desired by his cartel. In any case, a serious scholar would have requested clarifications to the author or to one of the several laboratories identified in the paper. Mr. Calo intentionally did not follow this standard "scientific process" for the evident intent to serve personal interests against undesired advancement of human knowledge and against the image of Brown University.
There are many serious misinterpretations, and misunderstandings
of the 'data" presented in this manuscript. Below are
just some of the most obvious that I encountered in the author's
efforts to establish "HHO gas" as a new form of matter.
Most of the correct interpretations of the data are quite straightforward, and certainly do not require the invocation of any new
form of matter.
p. 1114, line 56 et seq.: "Feature 1 of HHO gas:" The author
begins with the so-called "efficiency" of the electrolyzer,
E. The study of the physical behavior of water and water vapor
has been the subject of countless experiments over almost
a century, culminating in, among other things, the International
Steam Tables (1936). The latter include the energy required to
vaporize liquid water, or the enthalpy change of vaporization
of water. These values are very well known, and can also be
accurately measured in the laboratory with a calorimeter.
55 SCF (@ 60 F, 1 atm) of water vapor is about 1.18 kg.
The energy required to vaporize 1.18 kg of saturated liquid
water at 25 C is 1.18 kg 2442.3kJ/kg (enthalpy change
of vaporization of water @ 25 ?C) =2882 kJ. To take it to
saturated water vapor (or steam) at 35 psig requires about
another 184.5 kJ/kg, or an additional 218 kJ for 1.18 kg of
water, for a total of about 3.1 MJ. The author states that the
electrolyzer requires 5kWh or 18 MJ to vaporize the same
amount of water. ..... the entire argument about comparisons of efficiencies is
meaningless .....
In this arrogant show of old knowledge, Mr. Calo confirms his intentional scheme against society and against the image of Brown University. To begin, I stated quote clearly in Ref. [6] that my paper was devoted to the study of the gas produced by the electrolyzer of Hydrogen Technologies Application, Inc, and I merely provided efficiency data indicated by the manufacturer. Thus, entering into efficiency aspects on which I am not responsible is clearly
unethical. Independently from that, the above statement confirms Mr. Calo's dishonesty because in paper [6] I stated and repeated that the HHO gas is not expected to be a stochiometric mixture of H2 and O2 because said stochiometric mixture is known not to be able to melt bricks and tungsten instantly at contact. Therefore, I tentatively suggested that the HHO gas contains a yet unknown percentage of dimers OH and magnecules besides conventional; molecules, under which premises the above arrogant show off with old knowledge is clearly inapplicable, evidently because his old knowledge solely applies to the separation of water into H2 and O2 and not to the new chemical structure proposed for study in paper ]6]. The study of this new separation of water has been jeopardized and halted to this day by the scientific obscurantism imposed in the sector by Mr. Calo and his cartel via attacks and vexations on serious scholars working in the field. In any case, Calo's disqualification of Hydrogen Technologies Applications, Inc. (HTAI), without any direct inspection of its electrolyzer is so unethical, to provide amply grounds for the injured company to sue Brown University.
p. 1114, line 66: Consequently, the ensuing statement, "The
above efficiency establishes the existence of a transition of
water from liquid to the gaseous state that is not caused by
evaporation" has no basis in fact. The remainder of the paragraph
then jumps to the totally unsupported conclusion that
"HHO" must be another novel form of gas.
The use of this statement of my paper to attempt its disqualification is truly preposterous. Even high school students - except Mr. Calo - know that the gaseous state of water is not combustible, while paper [6] is devoted to the "combustible" character of the gas produced by the electrolyzer of HTAI.
p. 1114, line 74 et seq.: "Feature 2 of HHO gas:" The argument
about "oxygen depletion" is specious. The global oxygen
cycle is very well-known, and it maintains the concentration of
oxygen at sea level at a constant 21 mol%. Thus, it is quite an
exaggeration to imply that "oxygen depletion" is an environmental
problem.
A peculiarity of the HHO gas is that it burns without any need of atmospheric oxygen with consequential clear environmental values only enemy of society can deny. Brown University is alerted here because, in the absence of punitive action against Mr. Calo, said university will formally endorse anti-environmental misconducts since nobody - except Mr. Calo - can deny the environmental value of a combustible gas that requires no atmospheric oxygen. Moreover, Mr. Calo's dishonesty is further proved by the turning of my tentative words in the paper, such as "we expect ..." into Mr. Calo's twisting of "must". What a dishonesty!
p. 1114, line 84 et seq.: "Feature 3 of HHO gas:" The author
states that liquid water can be condensed out of "HHO gas"
at about 150 psi, while a much greater pressure is required to
do the same from "conventional gases". I really have no idea
what this means, but apparently the author has not considered
the phenomenon of rain. Water vapor in the atmosphere condenses
into liquid water at a total pressure of about an atmosphere
at sea level. It is all a question of humidity (or water
content or vapor pressure), and is very well-known. But, once
again, the author's incorrect statement and misunderstanding
lead him to conclude that "HHO gas" "does not possess a conventional
structure". Well, if that is so, then air must not possess
a "conventional structure" either. Once again, the argument
is specious, and the conclusions that the author jumps to are
completely unsupported.
The above statement is an additional proof of Mr. Calo's dishonesty because the thesis submitted in the paper with cautious words is that the HHO gas is "not" a stochiometric mixture of H2 and O2, but contains an unknown percentage of anomalous substances under which conditions the above pompous statement is inapplicable, as any serious scholar is expected to see and admit as a condition to qualify as such.
p. 1114, line 93 et seq.: "Feature 4 of HHO gas:" The author
cites "anomalous adhesion (adsorption) to gases, liquids,
and solids . . .," presumably of "HHO gas." I cannot even begin
to speculate on what "adhesion" of a gas to another gas
means, since adhesion requires the interaction of a species with
a bulk phase. However, vapor or liquid water is well-known
to "adhere" and adsorb onto many solids, which makes it an
ubiquitous contaminant in many such instances because of this
property. With respect to liquids, water can be absorbed in to
the bulk liquid phase of certain compounds, and can even concentrate
at liquid interfaces when surfactants are present. Since
the "adhesion" properties of "HHO gas" do not appear to be
any different than water vapor, how does this confirm a "novel
chemical structure" for "HHO gas," as claimed by the author?
Once again, the author cites a phenomenological observation,
misinterprets it, and comes up with an unwarranted conclusion.
This additional pompous review of old knowledge is highly unethical, arrogant and damaging to Brown University. The gist of the statement is that Mr. Calo knows everything and, therefore, in the event a claim of new knowledge is made, the claim is wrong or fraudulent.In fact, Mr. Calo dismisses the anomalous adhesion of the HHO gas on the basis of the lack of his knowledge and understanding, while computer chips are nowadays produced with the anomalous adhesion of a suitably treated protective gas. What is unreassuring is that the manufacturer do not disclose the novelty of their treatment due to the internationally known scientific corruption in academia opposing in a coordinated way basic advance.
p. 1114, line 101 et seq.: "Feature 5 of HHO gas:" The
author states that "HHO gas" has "a widely varying thermal
content . . ." This is such imprecise language that it does not
convey any real meaning. Apparently, however, it is related to
the observation that an "HHO gas" flame is cool in ambient
air, but can transfer large amounts of energy upon contacting
solid objects. Once again, due to the lack of calculations, "hard
numbers," and specifications characterizing the gas mixture
composition and the flow rates, temperatures, pressures of the
produced gas, etc., it is difficult to make accurate calculations.
However, I can think of at least one alternative explanation for
this behavior that just involves water.
Mr. Calo has not inspected the original papers by Cannizzaro, Avogadro, Lavoisier and other founders of chemistry. In the event he had done so, he would have known that their historical discoveries were presented in a very tentative and preliminary way. In the event said founders of chemistry had to reach the utmost possible maturity of expression, the discoveries would not have seen the light of the day. I personally detected a large variation of heat transfer by the HHO gas from a flame in air which has a temperature of a few hundreds Farenheight to temperature of over ten thousands Farenheight degrees needed for the instantaneous melting of bricks and tungsten at contact. It is known that combustible gases with a molecular structure release the same thermal energy in BTU/scf under whatever combustion in air. By contrast, in paper [6] I reported, apparently for the first time, evidence on the existence of a combustible gas with a "variable" thermal emission depending on its combustion. I presented this evidence for the intent that qualified chemists would investigate it. Rather than participating in this needed scientific process, Mr. Calo tries to disqualify the process with dishonesty because without any technical counter-argument, zilch, thus implying that the reported evidence simply does not exist. What a dishonesty! I abstain from reporting the sequel of this "dismissal" because it is an arrogant and vacuous show of old knowledge that has no connection to the reported evidence.
To heat liquid water from
25 to 150 C (as cited on p. 1115, line 1 in the article) and
vaporize it will take about 2.6 MJ/kg. Burning hydrogen produces
about 242 kJ/mol hydrogen. Thus the amount of liquid
water required to absorb the entire energy of the combustion
reaction by evaporating liquid water is about 0.09 kg water/mol
hydrogen burned. At 150 C the volume of the gas is about
34.7 l/mol, while the volume of liquid water is about 1 cm3/g.
If the composition of the "HHO gas" is about 95% hydrogen
and oxygen in stoichiometric ratio, and 5% water vapor (as
mentioned in the article; see below), then the amount of hydrogen
in the gas would be about 0.633 mol H2/mol "HHO
gas." Complete combustion of this amount of hydrogen would
produce about 153 kJ/mol "HHO gas." This amount of energy
would heat and volatilize about 0.059 kg, or about 59 cm3 of
liquid water/mol "HHO gas." Thus, on a volumetric basis, all
you would need is 0.059 l/34.7 l or about 0.17% by volume
of liquid water droplets in the "HHO gas" to "absorb" all the
energy from the combustion of the hydrogen in the gas..
Again, this statement is simply preposterous and extremely damaging to Brown University. Incontrovertible evidence for the HHO gas, the Brown gas and similar gases available in the market is that they instantly melt bricks and tungsten at contact without any need for atmospheric oxygen. Equally incontrovertible is the evidence, confirmed by serious chemists is that a stochiometric mixture of hydrogen and oxygen "cannot" instantly melt bricks and tungsten at flame contact. Mister Calo avoids very carefully the quotation of these well established chemical realities and proffers a pompous, arrogant and vacuous show off with old knowledge that, again, have no connection whatsoever to the real issue: prove with old chemical knowledge how a gaseous form of water is, first of all, combustible and, secondly, melts bricks and tungsten instantly at contact at flame contact. In the event interested in maintaining a beautiful and renowned image, it is time for Brown University to acknowledge that the above behavior is scientific corruption by one of its employees that has to be admitted and addressed as such.
p. 1115, line 8 et seq.: The author states the "anomalous" apparent
measured molecular weight of "HHO gas" as 12.3 g/mol
(line 14). The author also states that the molecular weight of a stoichiometric mixture of H2 and O2 gas resulting from the
decomposition of water is 11.3 g/mol (line 17). It is not. The
molecular weight of a stoichiometric mixture of completely decomposed
water (i.e., 2/3 H2, 1/3 O2) is:
(2/3)2 + (1/3)32 = 36/3 = 12 g/mol.
The author even writes the equation "(2 + 2 + 32)/3 = 11.3"!
The answer is obviously 12; his arithmetic is incorrect. (What
is the compound P2? (line 15) I guess it should be O2�typo?
What is HBN2? (line 16) Another typo?)
All you need to obtain an apparent molecular weight of
12.3 g/mol is to have some water vapor present, as there must be
since the product gas originates from the electrolysis of liquid
water. In fact, all it takes is 5% water vapor to increase the apparent
molecular weight of the gas mixture to 12.3 g/mol from
12 g/mol. At atmospheric pressure this corresponds to about
38mm Hg of water vapor, which would be the equilibrium vapor
pressure of water in a warm (near ambient) gas mixture
(33 ?C).
This additional statement by Mr. Calo is additional evidence of scientific corruption to its extreme because spectral analysis of the HHO gas, clearly reported in the paper but intentionally ignored, establishes the absence of 5% water vapor in the HHO gas, thus confirming that the chemical composition of this gas is anomalous, that is, outside old chemical knowledge. This is the scientific evidence. The pompous, arrogant and vacuous statements by Mr. Calo have the sole value of supporting a cartel against undesired new knowledge, as established by the close collaboration between Mr. Calo of Brown University and Mr. Arthur Rubin of Wikipedia (see the section below).
p. 1115, line 27: This line actually contains one of the very
few correct observations in the entire manuscript there is indeed
an "additional heavier species" in the gas mixture (than
hydrogen, that is) it is just water vapor. Thus, this simple
molecular weight determination does not at all suggest any new,
anomalous gaseous molecular forms other than just normal water
vapor.
Again corruption, corruption and corruption by Mr. Calo. The paper presents very accurate IR scans of the HHO gas that do not show the well known IR signatures of the water molecules, which signatures are used in various technologies, including the microwave oven Mr. Calo may use every day. Therefore, the anomalous species contained in HHO cannot possibly be water vapor in the indicated macroscopic percentage. Assuming that there was indeed a possibility for water vapor to represent the macroscopic percentage of the anomaly in specific weight, any serious scholar would have personally conducted the test prior to venturing a corrupt statement. Brown University is an excellent college having some of the most advanced laboratories for spectral and other analyses. In the event Mr. Calo was a honest scholar, he should have secured sample of the HHO or Brown gas (available free of charge for testing) and do all the measurements prior to throwing evil dirt on undesired new advances to serve the interests of an evil ring against mankind.
p. 1115, line 37: Here the author is attempting to refer to a
"gas chromatograph" not a "Gas Chromatographer" (Fig. 1).
Even if it was referred to as a gas chromatogram it would still
be totally wrong. Fig. 1 is not a gas chromatogram at all (i.e.,
it is not a plot of retention times of various species through
a chromatographic column, as measured by a detector) it is
a mass spectrum, most probably obtained with a quadrupole
mass spectrometer. The spectrum appears to be a normal one of
hydrogen and oxygen gas together with some water vapor and
other background species that are typically present in vacuum
chambers. The ordinate is labeled incorrectly it should be
"Faraday Cup Current (1E-11 A)" and the abscissa should
be "m/e mass-to-charge ratio." How can the author not know
that Fig. 1 is a mass spectrum, especially since he includes
additional mass spectra in Figs. 10�12. This creates some doubt
as to whether he actually knows the difference between a gas
chromatograph (GC) and a mass spectrometer (MS).
I reported verbatim data and statements released by fully identified laboratories. Brown University should be made aware that said laboratories may lose their license in the far fetched eventuality that Mr. Calo would be correct, in which case lawsuits by the injured laboratories against Brown University would be inevitable. The consideration of this legal consequences of Mr. Calo's corruption is necessary for Brown University to implement due punitive action.
A stoichiometric mixture of H2 and O2 gas resulting from the
decomposition of water is 11.3 g/mol (line 17). It is not. The
molecular weight of a stoichiometric mixture of completely decomposed
water (i.e., 2/3 H2, 1/3 O2) is:
(2/3)2 + (1/3)32 = 36/3 = 12 g/mol.
The author even writes the equation "(2 + 2 + 32)/3 = 11.3"!
The answer is obviously 12; his arithmetic is incorrect. (What
is the compound P2? (line 15) I guess it should be O2�typo?
What is HBN2? (line 16) Another typo?).
Paper [6] contains evident garbling caused by the conversion (done by the publisher) of my tex file into Word. Also, due to the passing of one year between my sending corrected galleys and the actual publication of the paper, the publisher ended up publishing uncorrected galleys with garbling. This occurrence is also indicated in rebuttals [34,35] as well as in our achieves. In his fanatic fervor to attack undesired new knowledge to serve an evil ring, Mr. Calo cannot contain himself for using even transparent garbling as "evidence" of the lack of value of the paper. What a dishonesty!
p. 1115, line 37: Here the author is attempting to refer to a
"gas chromatograph" not a "Gas Chromatographer" (Fig. 1).
Even if it was referred to as a gas chromatogram it would still
be totally wrong. Fig. 1 is not a gas chromatogram at all (i.e.,
it is not a plot of retention times of various species through
a chromatographic column, as measured by a detector) it is
a mass spectrum, most probably obtained with a quadrupole
mass spectrometer....... The author talks about a "new
species" at m/e=33 of "nonmolecular nature." How could any
chemical species be of "nonmolecular nature?" What does this
term even mean? Even radicals, ions, dimers and clusters have
a "molecular nature." In any case, m/e = 33 is probably just
HO+2 formed by ion�molecule reactions in the MS ion source;
e.g.O+2+ H2 ? HO+2+ H............ What about the
much larger m/e = 28 peak, which the author ignores? Is
it also another new "nonmolecular" species? No, as anyone
who has ever looked at background mass spectra knows fully
well, it is either just residual N+2 in the vacuum chamber, or
background CO (CO+) outgassing from the vacuum system
walls.
I stated several times that, in the event I am free to assume whatever I wish, I can prove that the Sun is made up by a set of excited bees, But I am a scientist and, as such, I do not fabricate a set of farfetched ad hoc beliefs to support a preset aim. By contrast, Mr. Calo is a corrupt scientist abusing the credibility of Brown University to venture a clear set of ad hoc assumption to achieve his preset, that of disrupting the due scientific process
in the pursuit of new scientific advances.
And what about the very large "negative peaks" like at
m/e=34? How can you get peaks of negative ion currents? Why
are they negative? It looks like either an electrometer problem
or a classical baseline problem. The mass peaks at 16 (O+
) and 17 (OH+) are classical "fragment
ions" PRODUCED from neutral water vapor in the ion
source of the mass spectrometer. They will be present whenever
neutral water vapor is present; i.e., they are "fingerprints"
of neutral water vapor. They are definitely not present as radicals
in the neutral sampled gas. The author apparently does
not understand how a mass spectrometer ion source functions.
He concludes that O and OH radicals are present in the "HHO
gas." Even if they were, they would certainly disappear by
homogeneous and heterogeneous processes (reactions) well
before they could be ionized and reach the detector.
This is a clear self-documentation of corruption because Mr. Calo acknowledges a main point of paper [6], namely, the claim that the HHO gas contains radicals OH and other anomalous species in a stable form. Yet all preceding pompous, and arrogant shows of old knowledge were referred to a gas without radicals, thus confirming the inapplicability of the fanatic arrogance and, therefore, Mr. Calo scientific corruption because his "argument" has no scientific value since it refers to a gas basically different than that suggested for study. Serious scholar will also note my tentative languages when faced with Terra Incognita such as the HHO gas compared to the pompous and arrogant show of old knowledge by Mr. Calo. I abstain from the reproduction and comments on additional similar statements by Mr. Calo because a continuation of the scientific scam, that is, the admission of clear anomalous MS clusters in the HHO gas and then the fabrication of all sport of unverified beliefs to "explain" the anomalies via old knowledge. Anybody who thinks that this is a serious scientific conduct for a serious employee of a serious college such as Brown University would need medical examination, or admit being a member of Calo's anti-American ring.
What about the unexplained peak at m/e=40,
which is labeled as 100% (which is customarily reserved for
the largest peak in the spectrum)? What is it, and where does
it come from? Once again, there is absolutely no explanation.
A large m/e =40 peak is usually indicative of a large air leak.
This peak originates from argon (Ar+) in the air.
Brown University must face the reality that there is a limit of indecency in scientific conduct beyond which any tacit acceptance is complicity and, therefore condemnation by society. As high school students know - except Mr. Calo - air only contains traces of Argon. As such, positively, absolutely, incontrovertibly Argon cannot explain the macroscopic peak at 40 amu. This claim by Mr. Calo passes all limits of scientific decency, thus showing the real Mr. Calo is a member of an evil cartel against society perpetrated under the credibility and abuse of Brown University.
Figs. 10 and 11, which the author does acknowledge as mass
spectra, exhibit bizarre, unexplained features that cast doubts
on their validity and interpretation. In Fig. 10 there is a peak
at m/e = 5 that is 75% of the hydrogen signal, but in Fig. 11
which is also supposed to be another spectrum of "HHO gas"
there is absolutely no hydrogen (m/e = 2) (How can that be?
By the author's own claims, hydrogen makes up the majority
of "HHO gas."). There is also no m/e = 5 for that matter. The
author never even attempts to explain these very disturbing
anomalies. All the other peaks in Fig. 11 are also common
background vacuum chamber peaks. They show 15 (CH+
3 ),
Again, in the event the above statement by Mr. Calo were true, the laboratories that released the scans should see the termination of their license due to Mr. Calo irresponsible statement and, therefore, said laboratories should sue Brown University fore motional; injuries and financial damages. In reality, we are dealing with very serious laboratories that repeated the scans several times before releasing the results with signed reports. Again, again and again, the HHO and Brown gases are commercially available,. Therefore, any serious scholar can secure them and independently verify the results "before" throwing scientific dirt at new advances. Mr. Calo intentionally abstained from doing that in order not to preclude his evil aim of disrupting due scientific process on new knowledge undesired by his evil anti-American ring. As an example, the peak at 5 amu is true. Period. It also appears routinely in scans of Magnegas and other anomalous fuels. Calo's statement <> is the sublimation of Calo's corruption because <> are, first of all, admitted by Calo and, secondly, clearly explained in paper [6] via the new experimentally established Santilli magnecules. At the peak with 5 amu
we have reached such a point of scientific corruption that Mr. Calo cannot any longer come out with a series of hyperbolic assumptions to "explain the peak, such as the imaginary "air leak" and the like. None of these manipulations can provide a credible
explanation of the stable pean at 5 amu present in the HHO, Brown, magnegas and other gases in detectable, repeatable and macroscopic percentage. Only Santilli magnecules, such as (H-H)x(H-H)xH can. This interpretation is the only available quantitative one published in refereed journal. Therefore, it is the scientific reality of the moment. Mr. Calo's posturing is just scientific fraud.
p. 1115, line 47 et seq.: The author talks about a "new
species" at m/e=33 of "nonmolecular nature." How could any
chemical species be of "nonmolecular nature?" What does this
term even mean?
In this statement Mr. Calo surpasses the dishonesty of preceding statements because he intentionally abstained to at least glancing at the preceding literature on he new species of Santilli magnecules, such as Refs. [1-5]. Yet, Mr. Calo uses his ignorance of new advances to dismiss them for clear personal gains. Particularly dishonest is Calo's statement that anomalous signatures in the blanks after tests following the flushing of the instrument are due to impurities of of preceding scans. What a vulgar dishonesty this is because mr. Calo knows that no laboratory can conduct and release scans without first checking that the equipment is free of contaminants. Yet in his fanatic fervor to attack unjustly and without provocation, as typical of his evil anti-American ring, he reaches the most absurd assumptions. I omit reporting additional "argument" by Mr. Calo in the above points in order not to abuse the serious reader.
p. 1116, line 9: Based on his incorrect interpretation of the IR
spectra, the author concludes that the m/e =18 peak observed
in the mass spectra ". . . is not water vapor as conventionally
understood in chemistry." However, the 18 peak is most certainly
nothing but "conventional" water vapor. How could the
"HHO gas" NOT contain water vapor? It is produced from
liquid water and thus is undoubtedly saturated with water vapor.
The above statement is the climax of Mr. Calo scientific corruption. In all serious analytic laboratories, "two" independent chemical analyses are made prior to any identification of a species. As an example, forensic laboratories are requested by law to use GC-MS as well as IR scans for the same species as a condition to prevent sending innocent people to the electric chair. Thank God, Mr. Calo has no such serious duties in society and so he pompously and arrogantly claims that a peak at 28 amu must necessarily be that of a water molecule. To support his dishonesty, he claims that the alternative interpretation very tentatively presented in paper [6] in serious scientific language is due to <>. That is a statement that requires the termination of employment by Mr. Calo by Brown University because the IR signatures of the water molecule are very well known. Since these signatures did not appear in the IR scans of HHO, then Mr. Calo claims that I had the "wrong interpretation of the IR scans" but he does not tell the - for him alone - "correct" interpretation of the IR signatures that would explain then pean at 28 amu as being that of water. he cannot and he knows it because he knows well his dishonesty and does not care because perpetrated against an outsiders of his evil anti-American cartel. In the absence of the IR signature of water, the peak at 18 amu "cannot" be due to the water molecule. Period. The same scientific corruption occurs for all anomalous species in HHO. According to incontrovertible chemical knowledge, lack of IR signature can only occur for perfectly spherical species. Also according to incontrovertible knowledge, peaks without any IR signature cannot be cluster of atoms under the conventional valence bond because such a bond will inevitably produce an IR signature due to their strength. Therefore, clearly identified macroscopic peaks in the MS which do not have IR signatures at their amu value cannot possibly be conventional molecules and, therefore, they constitute evidence of the new chemical species of Santilli magnecules. In addition to the denial of this evidence, there is an additional scientific corruption that has to be identified and denounced as such. When faced with macroscopic peaks in the MS that have no IR signature at their amu value, corrupt guys (I cannot possibly call scientists) immediately provide the IR signatures occurring at smaller amu values. This is scientific corruption because the IR signatures are those of the "constituents" of the cluster considered, and not of the cluster itself, thus bypassing the novelty of the bond at the amu value of the cluster for the clear intent of maintaining old knowledge against the interest of society. I strongly recommend Brown University not to back these manipulations of scientific evidence for evil interest.
p. 1116, line 13 et seq.: The author apparently does not understand
what bubbling an insoluble gas through a liquid hydrocarbon
fuel like diesel does. It is called stripping. Diesel
fuel is composed of many hydrocarbon compounds of varying
volatility. By bubbling "HHO gas" (which is primarily insoluble
hydrogen and oxygen) through the liquid diesel fuel,
there is preferential stripping (i.e., removal into the gas phase)
of the more volatile (i.e., higher vapor pressure) constituents
from the diesel fuel. Obviously, the flash point of the resultant
liquid fuel must then increase. So the result is anything but
anomalous, it is what would normally be expected. In fact, the
author concludes in a comparison of the data from the original
fuel and that after stripping, that there is "a bigger elution time,
and, above all, a shift of the peaks toward bigger amu values."
(p. 1117, lines 31, 32). He also goes on to say (p. 1118,
line 5) that the stripped diesel fuel clogged "the GC feeding
lines." This is all very good proof that the resultant hydrocarbon
mixture becomes heavier after stripping, which means that the
flash point must have increased. Thus the author does not interpret
the data properly, even when it shows the correct expected
behavior!
This statement is the assassination of dignity or shame on issues pertaining to our environment. In fact, Mr. Calo uses and abuses old knowledge and the credibility of Brown University to discredit, jeopardize and disrupt the development of new clean burning fuels consisting of a magnecular bond of conventional fuels with HHO, or other gases possessing magnecular structure, which development is under way following millions of dollars of funding. Confirmation of Mr. Calo's extreme corruption is his preceding denial of the evidence that blanks after flushing the instruments show macroscopic peans of the tested species as being due to fraud by the laboratories, rather than the admission that they are incontrovertible evidence of an anomalous adhesion of HHO, Brown, magnegas and other gases with magnecular structure. Officers of Brown University is suggested to ponder on the implication for their college as well as for the environment in the event of tolerance of such a blatant anti-scientific conduct. Similar incredible corruption is established by Mr. Calo denial of the clogging of microscopic feeding line by many gas with magnecular structure which is established in analytic laboratories in two continents. Flat denial of new evidence without any verification is indeed vulgar corruption against society. Period. In reality, basically "new" fuels are under development following large industrial investments, where the novelty is the new non-valence bond between the fossil fuel and the additive, as one can see, for instance, in the web site of Magnegas Corporation (www.magnegas.com). Brown university must be made aware that the above statement by Mr. Calo can be construed as implying that the technologies under development by a U. S. publicly traded company are are fraud, this mandating all necessary lawsuits by the stockholder for the projection of their image as well as the value of their stock. This Open Ethical Complain with Request for Punitive Action has been filed by the underwriter in the hope of "preventing" such legal actions in the only possible way: by terminating Mr. Calo as an employees of Brown University.
Thus, these data provide no basis whatsoever for hypothesizing
"the existence of an anomalous bond between the diesel
and the HHO gas . . .". The data behave as expected. A similar
result would have been obtained if the diesel had been stripped
with any insoluble gas, such as nitrogen, argon or helium.
The article is replete with "pseudo-science" descriptions and
explanations. One particular example appears in the caption of
Fig. 9:
Fig. 9. One of the anomalous blanks of the GC-MS scans
made by Toxic LTD Laboratories following the tests of the
HHO. The blank is firstly anomalous because only the background
should have been detected, thus indicating a bond
between the HHO gas and the walls of the instrument, whose
most plausible explanation is the magnetic polarization by
induction of said walls by a form of magnetic polarization of
the species composing the HHO gas. The second reasons for
the anomalous nature of the blank is that the substances detected
cannot possibly exist in the HHO gas produced from
distilled water, thus showing an accretion of bonds to the
instrument walls.
What does this even mean? Scientific and technological
language should be precise. However, this language is
obfuscatory�perhaps even intentionally so.
Again Mr. Calo demonstrates his preposterous and arrogant posture of dismissing technical statement on the sole grounds that he does not know them! This is absolutely preposterous and undignified for Brown University. In the event mr. Calo minimally qualified as a "scientist" he would have at least glanced at the post Ph. D. monograph [4] published by Kluwer Academic publishing and quoted in paper [6] among several others. But Mr. Calo felt no need of learning the new field of chemistry underlying paper [6] because, in his pompous mind, he knows everything and, as such, he does need to study nothing. What an assassination of the most elementary sense of scientific dignity and due process! I again abstain form quoting and commenting on additional statements by Mr. Calo in the issue in order not top offend the serious scholar.
The remainder of the manuscript is a discussion of the
author's theory of "magnecules" as related to "HHO gas."
"Magnecules" are evidently supposed to be something like radicals
bound to stable species by "opposing magnetic polarities
originating from toroidal polarizations of the orbitals of atomic
electrons." This is "pseudo-science" as well. H, O, OH, CH,
as well as a number of other radicals, are present in all sorts of
reacting systems. In flames they are the "chain carriers" for the
hundreds of chain reactions that occur. The kinetics of these
reactions has been studied in considerable detail and there are
very robust flame kinetics models available that predict flame
chemistry quite well. Due to the very reactive nature of these
radicals, they react very rapidly with stable molecules, usually
with little or no activation energy barrier. There is absolutely
no evidence presented that they could form long-lived "cluster"
species with stable molecules like water. If the author wishes
to establish the existence of such "radical cluster" species,
he should avail himself of the many laser spectroscopic techniques
that have been so well developed for the study of
radical species in combustion environments. Resultant spectra
of "radical cluster" species would be a good start in attempting
to establish the existence of anything like "HHO gas."
At this point, Brown University is faced again with its real responsibility vis-a-vis the numerous investors and thousands of stockholders of the American company Magnegas Corporation traded at NASDAQ under the symbol of MNGA because the very name of the company "Magnegas" Corporation, originated from the new chemical species of Santilli Magnecules and the above dismissal without any inspection of the experimental evidence as done by Mr. Calo would be extremely damaging to Brown University in the event Mr. Calo is maintained as an employees. In this case Brown University would be directly involved in dubbing as "pseudoscience" the main business of the U. S. public company Magnegas Corporation. Particularly damaging to Brown University would be the evidence in the use of magnegas fuel in various countries proving the fraudulent character of Mr. Calo's statements. For instance, according to Mr. Calo old knowledge, magnegas should have 315 BTU/scf , while magnegas cuts a 12" thick steel place faster than acetylene that has 2,400 BTU/scf. Denying novelty under this evidence is a scientific crime. To set the difference between me and Mr. Calo, I recall that, at the end of an invited seminar on magnegas I delivered at the Institute for Ultra Fast Spectroscopy of the City College of New York(see the DVD on the lecture
Lecture IV A:
An Introduction to Hadronic Chemistry,
Invited Lecture delivered at the
Institute for UltraFast Spectroscopy and Laser
City College of New York,
October 19, 2012 was asked" Prof. Santilli, what is magnegas?" I answered: "In my capacity [of the time] as Chief Scientist of a U. S. public company I have to state that I do not know." The admission of lack of knowledge is science. Every statement by Mr. Calo in his allegations [28] is the assassination of science the studious dismissal of novelty via the use of totally inapplicable old-decrepit knowledge belonging to the past millennium. In its decision, Brown University is suggested to show awareness that we are in the third millennium and that America need "new," "new," "new" knowledge to resolve our biggest deficit in the history of mankind and, therefore, individual such as Mr. Calo are a threat to the future of our country.
Conclusion
In conclusion, the author provides absolutely no scientific
evidence that supports the existence of a new form of matter
called "HHO gas." From the data presented, the gaseous product
from the electrolyzer behaves in the same manner as would
be expected of a mixture of hydrogen, oxygen, and water vapor.
The assassination of serious science perpetrated under the credibility of the otherwise beautiful and renowned Brown University reaches its climax in the above conclusion by Mr. Calo because he studiously ignores the anomalous combustion of the HHO gas in instantly melting bricks at contact without any need of atmospheric oxygen, Absolutely, positively, unquestionably none of the numerous mambo jumbo proffered by Mr. Calo in comments ['28] explains even minimally this incontrovertible anomaly that anybody can verify since the HHO gas is sole and used to the public in more than one continent. The interpretation of the HHO gas that I presented in paper [6] does indeed provide at least a tentative interpretation that should be subject to serious "quantitative" dismissals or confirmation to receive in return attacks of all kinds. Since the dismissal of the pursuit of new knowledge rather widespread, I want to close these comments by stating that, unless this trend is reverted and due scientific process based on counter-measurements or quantitative disproofs are implemented, particularly in the event back up via inaction by distinguished colleges such as Brown University, America may well risk entering into the dark side of scientific history.
3. DOCUMENTATION OF ORGANIZED ANTI-AMERICAN CONSPIRACY
IN CHEMISTRY INITIATED AT BROWN UNIVERSITY
As reviewed in Section 2, without any advance consultation or contact with me, the "engineer" Mr. J. M. Calo releases his unethical "criticisms" [33] under the official affiliation to Brown University which "criticisms" have been denounced and documented in Section 2.
As also recalled in Section 2, immediately following the appearance of "criticisms" [33] "Professor of Chemistry" M. Coleman from Ireland released his rebuttal [34] and "Professor of Chemistry" J. V. Kadeisvili from Georgia, Russia, released his own rebuttal [35], which two rebuttals are an integral part of this Ethical Complaint against Mr. Calo with Request of Termination of his Employment at Brown University.
In this section, I present a number of, direct and indirect, documentary evidence in the existence of an organized nation-wide anti-American conspiracy in chemistry that can only be denied by the inept or by the accomplice. I denounce elsewhere organized conspiracies in other field of physics.
Calo's conduct is fully in line with the "criticisms" I suffered when I was at Harvard University in the early 1980s under a formal grants from the Department of Energy. These "criticisms" were proffereed by Harvard's employees Steven Weinberg, Sheldon Glashow and (the late) Sidney Coleman who had such an unethical audacity to oppose the will of the Government of the United States of America by publicly dubbing my research under formal DOE grants as having "no physical values"
(see my 1984 denunciation Ethical Probe of Einstein Followers in the USA: An Insider's view, its three volumes of documentation and the supporting editorial of The Harvard Crimson dubbing these unethical conducts The Politics of Science). I accept and respect politics in politics, but I consider politics in science a true crime against society.
The internationally known and denounced reason for such an unethical obstruction by Harvard's employees Weinberg, Glashow and Coleman is that I had been requested by the DOE to study coverings of 20th century theories allowing a quantitative representation of energy releasing processes. These processes are notoriously irreversible over time (their time reversal image violates causality and other laws). Said employees of Harvard University opposed my research because Einstein theories are strictly reversible over time and, therefrom, they are "inapplicable" to irreversible processes (it would be unethical to use the word "violated" because Einstein conceived his theories solely for reversible processes).
Technically, the reversibility of 20th century theories - including that of Einstein special relativity quantum mechanics and quantum chemistry - is due to their Lie algebra structure is invariant under anti-Hermiticity. As a member of the Department of Mathematics at Harvard University, I had been requested by the DOE to study the so-called Lie-admissible covering of Lie's theory because it breaks said invariance, thus assuring a representation of irreversible process. I was also asked to study covering theories for energy releasing processes with a Lie-admissible structure, resulting in a necessary covering of Einstein theories, quantum mechanics and quantum chemistry. Weinberg, Glashow and Coleman are notorious fanatics of Einstein and, therefore, they openly opposed the construction of a covering of Einstein theories by solely showing interest in their academic and other greed without any sense of dignity or shame, since they went to the extreme of withholding my salary from my own DOE grant
for one entire year despite their knowledge that I had a family with children (then) in tender age to feed and shelter.
Following my 1984 public denunciation of the misconduct of the public discredit of my DOE research by the indicated employees of Harvard University, I thought that I would be left in peace to conduct the research I wanted. In reality, I was the victim of simply unbelievable attacks and vexations by a world-wide cartel composed by Weinberg-Glashow-Coleman follows, all attacks and vexation being against my pursuit of suitable coverings of Einstein's special relativity, quantum mechanics and quantum chemistry, despite these covering being
necessary for energy releasing processes, thus necessary for true environmental advances (see my
2007 denunciation Update and the
Denunciation of the Gangsterism by the physicist Steven Weinberg).
The full alignment of the misconduct by Mr. Calo, as employee of Brown University, with the immoral conduct by the employees of Harvard University Weinberg, Glashow and Coleman is simply incontrovertible. In fact, one of the central insufficiencies of quantum chemistry is that it is a strictly reversible theory, with no arrow of time beginning in its axioms. Consequently, quantum chemistry is fundamentally insufficient for the resolution of our alarming environmental problems because the latter are all due to energy releasing processes that are structurally irreversible. Of course, numerous important studies exist of chemical reactions via quantum chemistry, but they are essentially phenomenological, that is, observations of nature without a quantitative interpretation of the structure of the process. With his "criticisms" of my paper [6], Mr. Calo did directly and indirectly oppose studies on the generalization of quantum chemistry into the covering hadronic chemistry which I conceived precisely with an irreversible Lie-admissible structure, the reversible case being a trivial particular case with the irreversible Lie-admissible structure (see monograph [4] for technical details).
Following the appearance of "criticisms" [33], Mr. Arthur L. Rubin et al. of Wikipedia immediately reported Calo's immoral "criticisms" in my article at Wikipedia
https://en.wikipedia.org/wiki/Ruggero_Santilli with the statement
In 2006, Brown University Professor of Engineering Joseph M. Calo wrote in the International Journal of Hydrogen Energy that Santilli's article had "many serious misinterpretations, and misunderstandings of the 'data' presented", provided "absolutely no scientific evidence" to support HHO gas's existence and he described Santilli's Magnecules as "pseudo-science as well".[18](Ref. [33] here).
The full cooperation, and therefore conspiracy between Mr. Calo at Brown University and Mr. Rubin at Wikipedia is first established by the fact that Mr. Rubin systematically removed the quotation in my article of rebuttals [34,35] published in the same journal as that of unethical "criticisms" [33], in clear violation of Wikipedia rules, by therefore turning the "free" dubbing of Wikipedia a real shame for America.
Also, while emphasizing Mr. Calo as a "Professor of Engineering" venturing "criticisms" on post Ph. D. chemical topics (based on new mathematics of which Mr. Calo does not even know its name), Mr. Rubin prohibited the quotation in my Wikipedia article of publications [17,18] by "Professor of Chemistry" Don Shillady of the Department of Chemistry of Virginia Commonwealth University, and perpetrated other ethical misconducts denounced in separate complaints (se, e.g.,
http://www.scientificethics.org/Wikipedia-corruption.htm and others) that confirmed Wikipedia as being a true national share for the United States ofAmerica.
Whether the duo Calo-Rubin did or did not obey order from a sinister anti-American cartel, their joint misconduct in discrediting my research via the abuse of the credibility of their institutions did fully succeed in achieving their intent of disrupting and actually paralyzing research in the new chemical species of magnecules and related clean burning fuels, as well as in paralyzing the research in the U.S.A. on an irreversible representation of energy releasing chemical reactions so much needed to solve our mounting environmental problems.
In any case, it is important to indicate that, in the absence of any quotation of Calo's "criticisms" by Rubin in my article at Wikipedia, I would not filed this Ethical Complaint. Therefore, the gravity of the case and the ethical as well as legal responsibility of Brown University rest in the Calo-Rubin conduct.
Among a long documentation of anti-American scientific conspiracy in the United States ofAmerica, I indicate:
s
A) Arthur Rubin and his controllers at Wikipedia have been under massive criticisms by the American as well as international community for scientific misconduct, by causing a perhaps irreparable damage to the image of our science in the world as well as in history. This is one of countless examples, I quote the denunciations of 333 complaints files against Mr. Rubin at Wikipedia one can see in the DVD
http://www.youtube.com/watch?v=O6QA9VEpVjw. But, despite this massive criticisms of so blatant misconducts by Rubin and transparent violation of Wikipedia rules, Mr. Rubin is still there and continues his anti-American action without any sense of dignity or shame. Therefore, any person who does not see the existence in Wikipedia and maintaining Rubin in his unethical misconducts, that person is either inept or an accomplice.
B) The coordinated rejection by various journals of the American Chemical Society of rejecting "all" papers in hadronic chemistry submitted by me and by independent chemists, rejections culminating with the recent rejection for "reviewing" comprehensive experimental paper [25] by independent chemists on numerous confirmations of the new chemical species of magnecules
Denunciation of organized Scientific Crime in the U.S chemistry, while paper [25] was readily published abroad in a reputable refered journals in its originally submitted version with very important praises by the references. The obvious unethical conduct by the ACS editors - obvious members of said anti-American cartel - is that paper [25] was "experimental" and, as such, could not be credibly rejected with the usual editorial mumbo jumbo. But the organized cartel had ordered the ACS editors to reject paper [25] hence, they had no other choice then suppressing science by avoiding its review.
C) Following the writing with me of the seminal papers [17,18] in which we achieved for the first time a numerically exact representation of the binding energies of the hydrogen and water molecules from first unadulterated principles of quantum mechanics. Don Shillady, Professor of Chemistry at Virginia Commonwealth University, was prohibited by evident member of the anti-American cartel at Virginia Commonwealth University to be a co-author of monograph [4] on hadronic chemistry, namely a theory in which he has been one of the main founder. Additionally, professor of Chemistry Don Shillady has been prohibited to participate at meetings presenting talks on "Santilli-Shillady strong valence bond" or on "Santilli-Shillady restricted three-body model of the hydrogen molecule" and has suffered prohibitions to be present at the defense of Ph. D. Theses in Chemistry carrying his name in the title, all these evidently in foreign countries due to the notorious impossibility of even consider a Ph. D. Thesis on chemistry in the United States of America on a covering theory for the representation of energy releasing processes. Any person who does not see and admit the existence of a capillary anti-American conspiracy in the United States of America is either an inept or an accomplice.
D) Following the appearance of dismissals [34,35], Chemistry Professor Cloonan became the victim of an organized harassment for his stand in Ref. [34] to such a degree that he was forced to terminate active research in the field following the important discoveries in Refs. [7-9] and while he was at the edge of achieving a new chemical species based on Santilli's hadronic chemistry.
E) Following the appearance of rebuttal [35], the late Chemistry Professor J. V. Kadeisvili was also the victim of such vexations and harassment expectedly by Mr. Calo's cartel, e.g., via the email thomas@flyingkettle.com and other emails that caused him severe emotional distress, since Prof. Kadeisvili lived all his life in a wheelchair due to serious birth defects, all this perpetrated with impunity to date in the United States of America.
In view of the serious misconducts surrounding this "Ethical Complaint with request for Punitive Action," A number of concerned scientists including myself has hired an Investigative Agency to ascertain and document whether: 1) the harassments and vexations suffered by Professors Cloonan, Kadeisvili and others, did indeed originate at Brown University and/or were commissioned by Mr. Calo under his official affiliation to Brown University; 2) Whether or not the scientific conduct by Mr. Calo of Brown University was indeed coordinated with the similar conduct by Mr. Rubin of Wikipedia, thus being conspiratorial; and 3) Whether or not Mr. Calo and Mr. Rubin are mere executors of orders originated by Mr. Weinberg and Mr. Glashow, thus establishing the existence in the United States ofAmerica of a sinister anti-American cartel potentially operating under billions of misused public funds. The results of the ongoing investigation will be publicly released at the time of our choosing.
In view of Mr. Calo blatant scientific corruption, the proffering of criticisms with complete lack of scientific value, the clear abuse of the credibility of the beautiful and renowned Brown University, the vexation and attack of qualified dissident scientists, the initiation of a scientific obscurantism in the sector, the clear damage to the image of America's science in the world, the coordination of his evil action with Wikipedia as separately denounced, personal emotional injury, and large financial losses, in my capacity as a pro se stockholder of Magnegas Corporation I hereby request that Brown University terminates the employment of Mr. Joseph M. Calo and provides other reparatory measures deemed appropriate.
I would like to close this ethical complain by addressing fellow American who are the real victims of the organized scientific scams by corrupt employees of our universities under the misuse of trillions of taxpayer money, and suggest fellow Americans to real in denunciation Denunciation of organized Scientific Crime in the U.S chemistry, my fear, by praying to God to be wrong, that America is doomed because of dimension, capillary and totally unchallenged character of the ongoing organized scientific corruption in our country.
The organized scientific crime in the U.S.A. has been abusing, for a century our money, jeopardizing our dignity and damaging our leadership in the world and in history. It is time for the silent majority to stop tolerating such an organized scientific crime and oppose it by following its structure, that is, via our own coordinated opposition. In its absence I have to remember to fellow Americans that , as written in history, people have the destiny that they either want or deserve.
REFERENCES
[1] R. M. Santilli, Theoretical prediction and experimental verification of the new chemical species of magnecules, Hadronic J. 21, 789 (1998)
http://www.santilli-foundation.org/docs/Santilli-43.pdf
[2] M.G. Kucherenko and A.K. Aringazin, Estimate of the polarized magnetic moment of the isoelectronium in the hydrogen molecule, Hadronic J. 21, 895 (1998),
http://www.santilli-foundation.org/docs/estimate.pdf
[3] A. K. Aringazin, Toroidal configuration of the orbit of the electron of the hydrogen atom under strong external magnetic fields, Hadronic J. 24,, 134 (2001), available in free pdf download from the link
http: www.santilli-foundation.org/docs/landau.pdf
[4] R. M. Santilli, Foundations of Hadronic Chemistry, with Applications to New Clean Energies and Fuels, Kluwer Academic Publishers (2001), http: //www.santilli-foundation.org/docs/Santilli-113.pdf.
[5] R. M. Santilli, The novel magnecular species of hydrogen and oxygen with increased specific weight and energy content, Intern. J. Hydrogen Energy 28, 177-196 (2003),
http://www.santilli-foundation.org/docs/Santilli-38.pdf
[6] R. M. Santilli, A new gaseous and combustible form of water, Intern. J. Hydrogen Energy 31, 1113 (2006),
http://www.santilli-foundation.org/docs/Santilli-138.pdf
[7] M. O. Cloonan,Application of the Cplex-isoelectronic theory to electrocyclisations, sigmatropic rearrangements, cheletropic reactions and antiaromaticity: Consistent with Santilli's hadronic chemistry. � Int. J. Hydrogen Energy 32 , 3026 (2006),
http://www.santilli-foundation.org/docs/Cloonan-Paper2.pdf
[8] M. O. Cloonan, A new electronic theory of pericyclic chemistry and aromaticity is proposed: The Cplex-isoelectronic theory. Consistent with Santilli's hadronic chem- istry,� Int J Hydrogen Energy, 32, 159 (2007),
http://www.santilli-foundation.org/docs/Cloonan-Paper1.pdf
[9] M. O. Cloonan, Origin of the endoselectivity observed in pericyciclic reactions based on the CPlex-Isoelectronic theory, Hadronic J, 32, 125 (2009),
http://www.santilli-foundation.org/docs/Cloonan-Paper3.pdf
[10] R. M. Santilli, The New Fuels with Magnecular Structure, International Academic Press (2008)
,
http://www.i-b-r.org/docs/Fuels-Magnecular-StructureF.pdf,
Italian translation available from the link
http://www.i-b-r.org/docs/Carb-Strutt-Magnecolare.doc
[11] R. M. Santilli, Experimental Confirmation of Nitrogen Synthesis from deuterium and Carbon without harmful radiations, New Advances in Physics Vol. 4, page 29, 2011,
http://www.santilli-foundation.org/docs/ICNF-1.pdf
[12] Robert Brenna, Theodore Kuliczkowski, and Leong Ying, Verification of Santilli in- termediate Controlled Nuclear Fusions without harmful radiations and the production of magnecular clusters,� New Advances in Physics, Vol. 5, page 9 (2011)
http://www.santilli-foundation.org/docs/ICNF-2.pdf
[13] R. M. Santilli, Additional Confirmation of the Intermediate Controlled Nuclear Fusion without harmful radiation or waste, in the Proceedings of the Third International Conference on the Lie-Admissible Treatment of Irreversible Processes, C. Corda, Editor, Kathmandu University (2011) pages 163-177
http://www.santilli-foundation.org/docs/ICNF-3.pdf
[14] R. M. Santilli, Isotopic, Genotopic and Hyperstructural Methods in Theoretical Biology, Ukraine Academy of Science, first edition (1997),
http://www.santilli-foundation.org/docs/santilli-67.pdf
[15] Day D. TCD analysis and density
measurements of Santilli Magnehydrogen. Eprida Laboratory report
dated 11/10/11.
http://www.santilli-foundation.org/docs/Eprida-MH-Certification-10-11.pdf
[16] Y. Yang, et al, Experimental Confirmations of Santilli's MagneHydrogen, Interna- tional Journal of Hydrogen Energy 38, 502 (2013)
http://www.santilli-foundation.org/docs/MagneHydrogen-2012.pdf
[17] R. M. Santilli and D. D. Shillady,, "A new isochemical model of the hydrogen molecule," Intern. J. Hydrogen Energy Vol. 24, pages 943-956 (1999)
http://www.santilli-foundation.org/docs/Santilli-135.pdf
[18] R. M. Santilli and D. D. Shillady, , "A new isochemical model of the water molecule," Intern. J. Hydrogen Energy Vol. 25, 173-183 (2000)
http://www.santilli-foundation.org/docs/Santilli-39.pdf
[19] R. Perez-Enriquez and R. Riera, "Exact analytic solution of the restricted three-body Santilli-Shillady model of the hydrogen molecule, " Progress in Physics Vol. 2, 34-41 (2007) (physics/0001056)
www.santilli-foundation.org/docs/3body2.pdf
[20] A. K. Aringazin and M.G. Kucherenko, "Exact variational solution of the restricted three-body Santilli-Shillady model of the hydrogen molecule," Hadronic J. Vol. 23, 1-56 (2000) (physics/0001056)
www.santilli-foundation.org/docs/3body.pdf
[21] E. Trell, Review of Santilli's Foundations of Hadronic Chemistry, Intern. J. Hy drogen Energy 28, 251 (2003),
http://www.santilli-foundation.org/docs/Trell-review-HC.pdf
[22] V. Tandge Hadronic Chemistry Applied to Hydrogen and Water Molecules, AIP Conf. Proc. 1479, 1013 (22012)
http://www.santilli-foundation.org/docs/Tandge-AIP.pdf
[23] Sangesh P. Zodape and Anil A. Bhalekar,
The Magnehydrogen in hadronic Chemistry
American Institute of Physics, in press 2013
http://www.santilli-foundation.org/docs/Zodape.pdf
[24] I. Gandzha and J. Kadeisvili, New Sciences for a New Era: Mathematical, Physical and Chemical Discoveries of Ruggero Maria Santilli, Sankata Printing Press, Nepal (2011),
http://www.santilli-foundation.org/docs/RMS.pdf
[25] Y. Yang, J. V. Kadeisvili, and S. Marton
Experimental Confirmations of the New Chemical Species of Santilli Magnecules,
The Open Physical Chemistry Journal Vol. 5, 1-16 (2013)
pdf available from the link
http://www.santilli-foundation.org/docs/Magnecules-2012.pdf
[26] International Conference on Hadronic Chemistry, Mathematics and Physics
October 21 to 26, 2013, Department of Chemistry
Rashtrasant Tukadoji Maharaj Nagpur University,India
http://www.nagpurtoday.in/2013-international-workshop-on-hadronic-chemistry-mathematics-and-physics-gets-underway-at-nu-campus/
[27] Prof. Santilli's Keynote Lecture at the
2013 Indian Conference on hadronic Chemistry:
An Introduction to Hadronic Chemistry
http://www.santilli-foundation.org/Santilli-India-2013.php
[28] Santilli IsoDifferential Calculus, IsoDynamical Systems and their Applications
Session ss56 of the
American Institute of Mathematical Sciences, Madrid, Spain, July 7 to 11, 2014
Organizers: S. Georgiev, C. Corda, and A. A. Bhalekar
http://www.aimsciences.org/conferences/2014/2014_specialSessions.html
[29] International Workshop on Santilli mathematics and their applications
to be held at the Session M3 of 2014 ICNPA Congress: Mathematical Problems in Engineering, Aerospace and Sciences, July 15-18,2014, Narvik University, Norway.
http://icnpaa.com/index.php/icnpaa/2014/schedConf/trackPolicies
[30] Fifth International Workshop on Santilli's Iso-, Geno-, and Hyper-Mathematics
Organizers: A. Bhalekar (India), C. Corda (Italy), and T. Vougiouklis (Greece)
http://www.santilli-foundation.org/Iso-Geno-Hyper-Math-2014.php
Session 108 of the ICNAAM meetings in Rhodes, Greece, September 22 to 216, 2014
http://www.icnaam.org/sessions_minisymposia.htm
[31] PRWeb news reklease
The New Chemical Species of Santilli Magnecules Confirmed
at the Recent ICNAAM Meeting in Greece
Announcement
http://www.prweb.com/releases/2013/10/prweb11208713.htm
Report
http://www.santilli-foundation.org/docs/PRWeb-No-6.pdf
[32] PRWeb News release,
Hadronic Chemistry, MagneHydrogen and Hy-Fuels Studied at an
International Workshop Organized by the Department of Chemistry of
R. T. M. Nagpur University in India
Announcement
http://www.prweb.com/releases/2013/10/prweb11233288.htm
Report
http://www.santilli-foundation.org/docs/PRWeb-No-8.pdf
[33] J. M. Calo, "Comments on: A new gaseous and combustion form of water," Intern. J.
Hydrogen Energy 32, 1309 (2007),
http://www.santilli-foundation.org/docs/Calo.pdf
[34] M. O. Cloonan, "A chemist view of J. M. Calo's comments on: A new gaseous and
combustible form of water," Intern. J. Hydrogen Energy33, 1309 (2008),
http://www.santilli-foundation.org/docs/Cloonan.pdf
[35] J. V. Kadeisvili, "Rebuttal of J. M. Calo's comments on R. M. Santilli's HHO paper,"
Intern. J. Hydrogen Energy 33, 918 (2008),
http://www.santilli-foundation.org/docs/Kadeisvili.pd
===================================
November 8, 2014
To Dr. Christina Paxson
Office of the President
and
Dr. Lawrence E. Larson
Dean of Engineering
with copy to
Mr. J. M. Calo
Division of Engineering,
Box D, Brown University
Dear Drs. Paxson and Larson,
This is to inform you of my uploaded but unlisted Ethical Complaint
DENUNCIATION OF SCIENTIFIC GANGSTERISM AT
BROWN UNIVERSITY WITH REQUEST TO TERMINATE
THE EMPLOYEE JOSEPH M. CALO
http://www.i-b-r.org/J-M-Calo-Denunc.htm
and request that, on or before thirty days from today, J. M. Calo
1) Apologizes to me in writing;
2) Publishes in the Comments of the International Journal of Hydrogen Energy the annul of his dishonest attacks with the statement
I hereby annul my comments
[1] J. M. Calo, "Comments on: A new gaseous and combustion form of water," Intern. J. Hydrogen Energy 32, 1309 (2007),
following the appearance of the following and other rebuttals
[2] M. O. Cloonan, "A chemist view of J. M. Calo's comments on: A new gaseous and combustible form of water," Intern. J. Hydrogen Energy33, 1309 (2008),
[3] J. V. Kadeisvili, "Rebuttal of J. M. Calo's comments on R. M. Santilli's HHO paper," Intern. J. Hydrogen Energy 33, 918 (2008),
and
3) Requests in writing with copy to me to Arthur Rubin, editor of my Article at Wikipedia the removal of the quotation of his dishonest attacks [1].
In the event the above requests 1, 2, 3, are met on or before thirty days from today,
I am at your disposal to sign a mutual release to your satisfaction prepared by your attorneys or mine.
Yours, Truly
===================================






